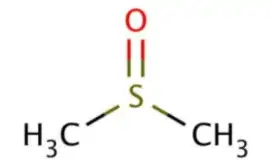
The Journey of Dimethyl Sulfoxide (DMSO)
Dimethyl Sulfoxide (DMSO): The Journey Of Dimethyl Sulfoxide, From Identification To Manufacturing
 Industrial-grade Dimethyl Sulfoxide (DMSO) has multiple applications in various spheres, increasing the demand for continuous manufacturing.
Industrial-grade Dimethyl Sulfoxide (DMSO) has multiple applications in various spheres, increasing the demand for continuous manufacturing.
The industrial manufacturing of dimethyl sulfoxide (DMSO) is an extremely lengthy and complicated process that includes several chemically verified and professionally executed steps.
So, let’s understand the chronology in detail.
Step 1: Identification and Detection Methods of Dimethyl Sulfoxide (DMSO)
The easiest method is to measure 1.5 ml of dimethyl sulfoxide, which needs to be slowly added to 2.5 ml of cooled hydroiodic acid by using a dropper.
The resulting solution is filtered and dried under reduced pressure along with the residue, usually a dark purple crystalline solid.
These resultants are unstable and have a distinctly unpleasant smell. Finally, dissolving the residue in chloroform forms a red solution.
Step 2: Dimethyl Sulfoxide (DMSO) Production Methods
We are already familiar with the process of oxidizing dimethyl sulfide to obtain this precious chemical compound. Depending on the type of oxidant used and the oxidation method followed, there is a classification in the production process. Some of them are listed below :
- Hydrogen Peroxide method – Acetone is chosen as the buffer medium where the reaction between hydrogen peroxide and dimethyl sulfide occurs.
- This process usually demands a higher cost than other known processes; hence, it is ideal only for large-scale production.
- Method using Nitrogen Dioxide – This is a relatively time-consuming synthesis method in which, on one hand, methanol and hydrogen sulfide are reacted in the presence of γ-alumina to obtain dimethyl sulfide. On the other hand, sulfate is reacted with sodium nitrate to generate nitrogen dioxide.
- Then, Dimethyl sulfide and nitrogen dioxide undergo a gas-liquid phase reaction at 60-80 degrees Celsius to produce crude dimethyl sulfoxide.
- Alternatively, Oxygen can be used for direct oxidation to obtain the same product. The final product is refined to obtain the good-grade DMSO once the vacuum distillation is over. This relatively advanced production method requires more time, effort and ingredients.
- Dimethyl sulfate method – In this process, dimethyl sulfate reacts with sodium sulfide to get the desired product. This has three sub-level reactions. Firstly, sulfate reacts with sodium nitrate to produce nitrogen dioxide.
- Secondly, dimethyl sulfide is oxidized with the resultant to obtain crude dimethyl sulfoxide.
- Finally, the crude variety undergoes neutralization and distillation to obtain superior dimethyl sulfoxide. Furthermore, the anodic oxidation method can help produce DMSO directly from dimethyl sulfide.
Step 3: Large-scale manufacturing process of Dimethyl Sulfoxide (DMSO)
After deciding upon one of the production methods, the full-fledged manufacturing process picks up pace at the industrial set-up.
To begin with, oxygen current at the rate of 370ml/min is bubbled through an approximately 30 cm layer of dimethyl sulfide between 26-27 degrees Celsius, which produces a gaseous mixture of oxygen that facilitates the oxidation process of the sulfide.
Then, nitric oxide is added to the obtained gaseous mixture at the rate of 30ml/min while it passes into an interconnected set of reaction chambers.
They are a series of 4 individually encompassing a glass tube of 4.3 cm in diameter and approximately 100cm in length.
Most of the reaction happens in the initial two tubes, and the temperature rises to 75°C, and gradually, the rate slows down in the final part of the chamber.
Post this, we observe a crude, yellow product dripping from the set-up. It has a composition of 10% DMSO, 2% nitrogen dioxide in dissolved form and minimum percentages of methane sulfonic acid added with water.
The obtained product is refluxed for 30 minutes at 100 degrees, and the resultant gas is passed through the first reaction chamber.
Finally, dimethyl sulfoxide is segregated by heating the product at extremely high temperatures while the other by-products are removed by adding slaked lime and vacuum distillation.
The above-stated breakdown is the methodology followed to yield pure dimethyl sulfoxide that makes its way for further use.